How To Apply QbD to Drug Device Combination Products – Quality by Design for Biotech, Pharmaceutical and Medical Devices
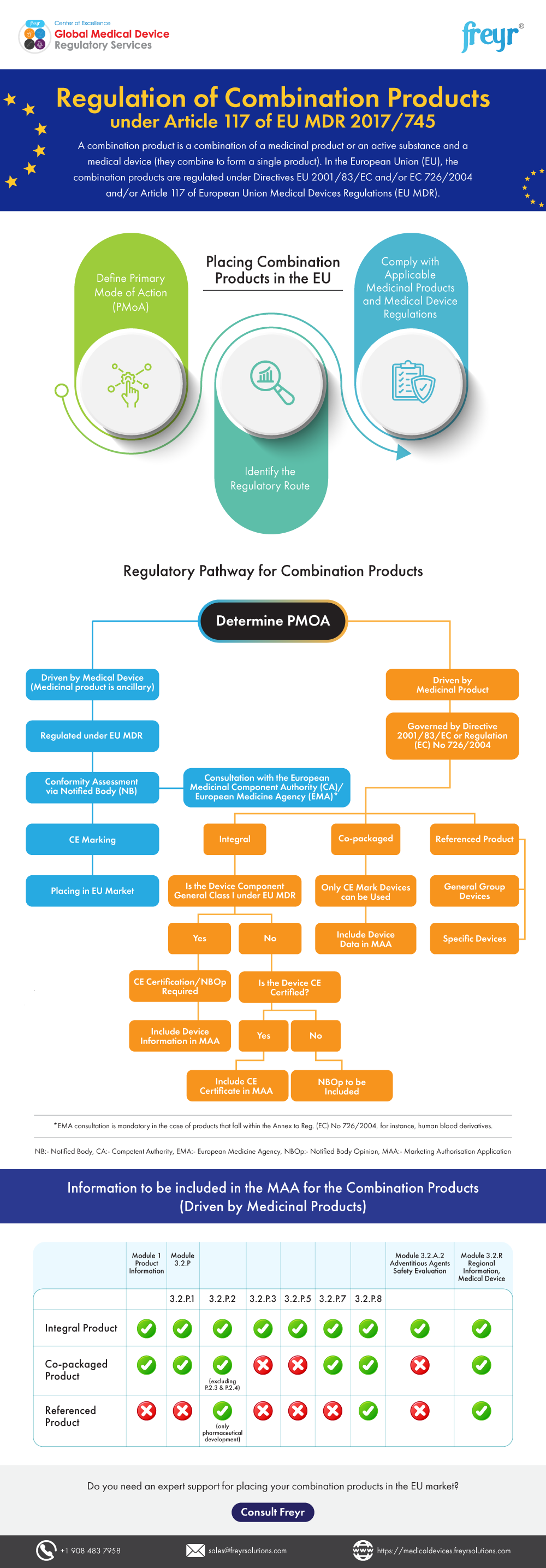
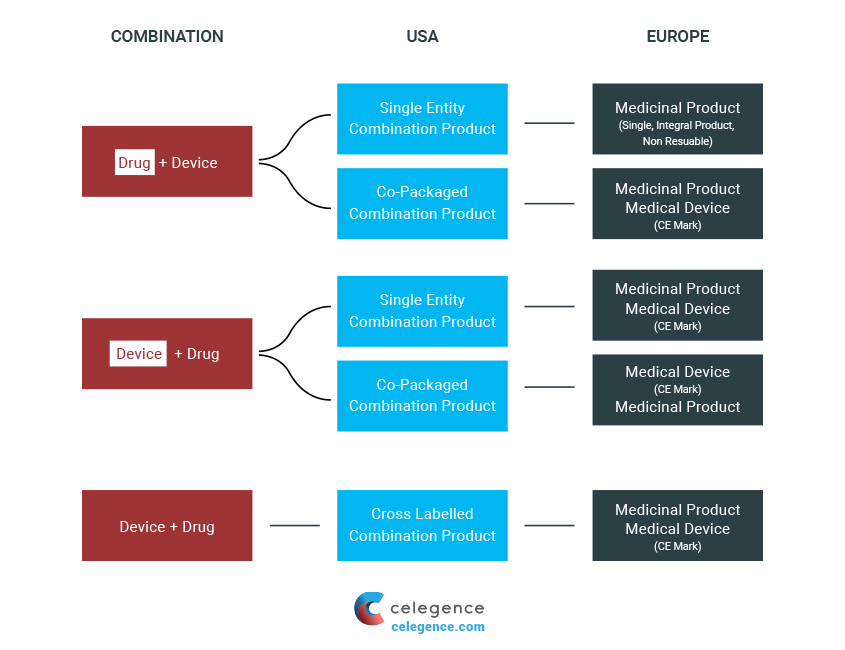
Regulation of Combination Products under Article 117 of EU MDR 2017/745 | Freyr - Global Regulatory Solutions and Services Company

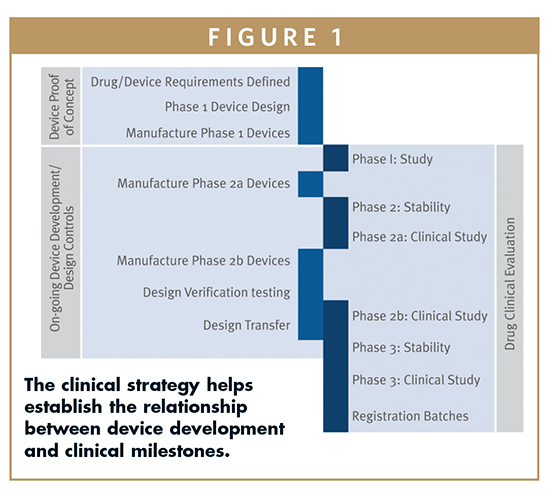
Product Development and Manufacturing Challenges for Combination Products | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Delivery to the eye: overcoming complex regulatory hurdles for combination products | Cambridge Consultants